
In Caenorhabditis elegans, twitchin is a giant polypeptide located in muscle A bands. The protein kinase of twitchin is autoinhibited by 45 residues upstream (NL) and 60
residues downstream (CRD) of the kinase catalytic core. Molecular dynamics simulation on a twitchin fragment revealed that the NL is released by pulling force. However, it is unclear
how the CRD is removed. To identify proteins that may remove the CRD, we performed a
yeast two-hybrid screen using twitchin kinase as bait. One interactor is MAK-1, C. elegans
orthologue of MAPKAP kinase 2.
The CRD of twitchin is important for binding to MAK-1 (mak-1 is expressed in nematode body wall muscle). Whereas unc-22 mutants are completely resistant, mak-1 mutants are partially resistant to nicotine. Genetic data suggest the involvement of two other mak-1 (mnk-1 y mak-1) paralogues and two orthologues of p38 MAP kinase (pmk-1 y pmk-3)
Nicotine sensitivity assays was made with synchronized young adult animals of each genotype collected in M9 buffer containing 0.01%Triton X-100 (M9T) For the assays, 96-well plates were used. 50–100 worms/well were added. The plate was placed in the dark at room temperature for 1 h. The measurement was made using the WMicrotracker One device. The locomotive activities under no-nicotine conditions were measured for 1 h . After the measurement, 50 μl of M9T containing 0.2 or 0.1% nicotine solution was added to each well . For each strain and nicotine concentration, eight independent wells were assayed. On the graphs of locomotive activity versus time, each point represents the mean and SE.
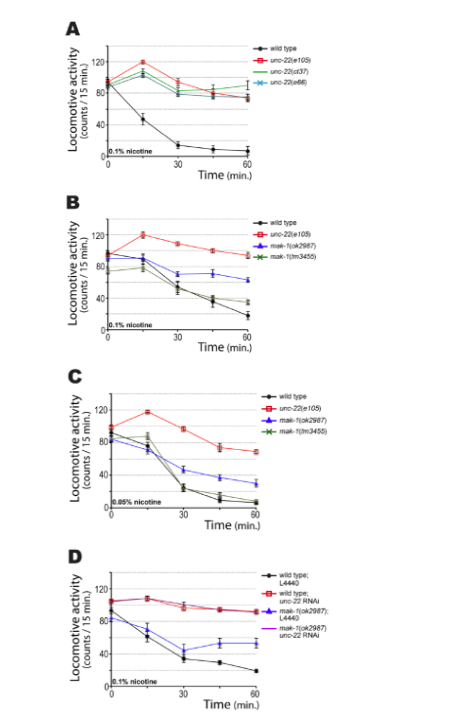
Figure 7 shows the response of wild-type, unc-22, and mak-1 mutant strains to nicotine. As shown in Figure 7A about 90% of the wild strain stops moving after approximately 30 min. In contrast, unc-22–mutant animals, regardless of allele, continue moving even after 60 min. In Figure 7B, the mutants mak-1 shows an intermediate response, that is, are partially resistant. The null mak-1 allele (ok2987) shows greater resistance than mak-1 (tm3445), and this effect is visible at nicotine concentrations of 0.1 or 0.05% (Figure 7C). Because both the unc-22 and mak-1 mutants show an abnormal response to nicotine, we wanted to analyze how the double mutant would respond. For this, the wild strain and mak-1 (ok2987) were fed with interfering RNA-producing bacteria that silenced unc-22.
As indicated in Figure 7D, the double mutant mak-1 (ok2987), unc-22 (RNAi) shows complete resistance, as does unc-22 (RNAi). That is, unc-22 is epistatic to mak-1.
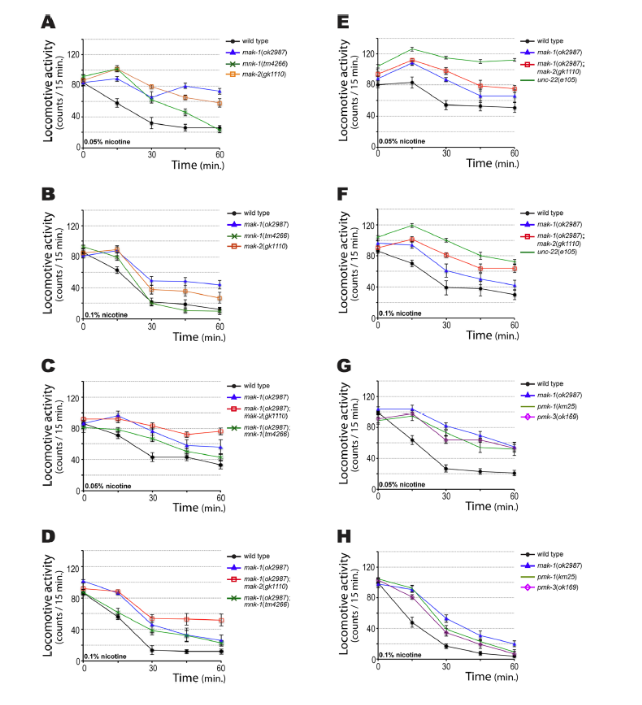
In figure 9, it can be observed that mnk-1, mak-2, pmk-1, and pmk-3 are partially resistant to nicotine.Using the same WMicrotracker assays, we found that mnk-1 and mak-2 are partially resistant to nicotine (Figure 9, A and B), and their resistance is in the order mak-1 > mak-2 > mnk-1. We also examined the nicotine response of the double mutants mak-1; mak-2 and mak-1; mnk-1. The mak-1; mak-2 mutant showed more resistance than mak-1 alone (Figure 9, C and D) but less resistance than unc-22 (e105) (Figure 9, E and F). These results suggest that mak-1 and mak-2 contribute to nicotine resistance in the same pathway, but other unidentified players are probably also involved.
As noted, mammalian MAPKAP kinase 2 is phosphorylated, and activated, by p38 MAP kinase. C. elegans has three p38 MAP kinases. Nevertheless, an intragenic deletion of pmk-2 is lethal in step L1, and prevents its analysis with the WMicrotracker registration system. However, as shown in Figure 9, G and H, the loss of function of pmk-1 or pmk-3 resulted in a partial resistance to nicotine, similar to mak-1.This result is consistent with the fact that pmk-1 and pmk-3 are part of the same genetic pathway as mak-1.
