
Angiostrongylus cantonensis, commonly known as the rat lungworm, is widely present across the humid tropics and subtropics as well as in a few more temperate regions and is the main aetiological agent of eosinophilic meningitis globally. The disease, known as neuroangiostrongyliasis, is a serious, sometimes fatal but neglected disease, not only in humans but also in domestic animals and wildlife. The limited number of anthelmintics available is concerning, especially because of increasing drug resistance. Anthelmintics are commonly derived from natural products, e.g. fungi and plants. This paper introduces the first use of the wMicroTracker system to evaluate the success of a large suite of natural products in arresting motility of A.cantonensis L3, the infectious stage of the parasite. The existing protocol for C.elegans was modified to assess inhibition of A.cantonensis L3 motility (and hence reduce infection potential) by a suite of bioactive natural products derived from the secondary metabolites of Hawaiian fungi.
Methods
Screening of natural product samples. Plates of 384 round-bottom wells were used; these concentrate the larvae in the centre of the well, within the path of the laser beams, and permit consistent and meaningful motility results. The optimum content of each well was determined to be as follows: ∼40 larvae (suspended in 19 μL water), 20 μL 1× phosphate-buffered saline and 1 μL dissolved library of natural products isolated from Hawaiian fungi (concentration 1 mg mL−1 for fractions, 0.1mgmL−1 for pure compounds, with the exception of M200 and M201), for a total well volume of 40 μL. For each sample, larval motility over the 72 h run period was plotted and compared visually to the larval motility in buffer (baseline motility), and subsequently categorized by their effect on motility: when motility was reduced to 0–25% of the maximum exhibited, the sample was categorized as high interest; when motility was reduced to 25–50%, as moderate interest; when motility was reduced to 50–75%, as low interest, and when motility was reduced to 75–100%, as no interest.
Results
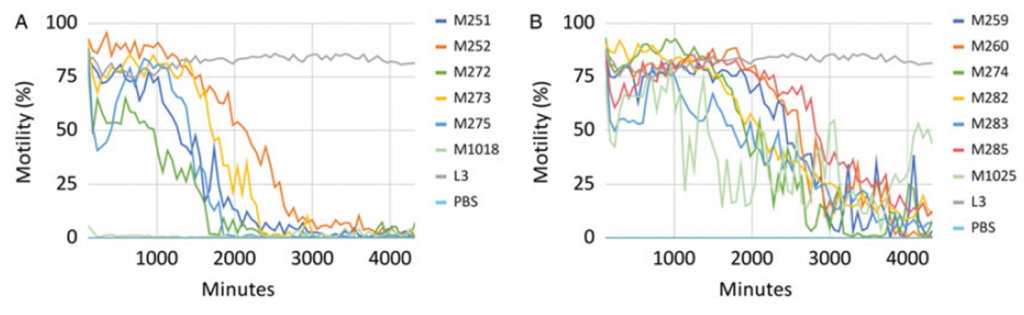
Using the optimized protocol for A.cantonensis, 108 samples (55 pure compounds along with their nontoxic parent crude extracts, fractions and sub-fractions) were screened, and 13 samples (two pure compounds and 11 fractions or sub-fractions) were identified as being in the high interest category (reduction to 0–25% motility). Of these 13 samples, five (M251, M252, M272, M273 and M275) caused essentially immediate and dramatic (close to 99%) motility reduction, which was sustained throughout the72 h period with little variability (Fig. 5A). These five samples (all were fractions) were considered of priority interest. Another six (M259, M260, M274, M282, M283 and M285) of the 13 high-interest samples were especially effective at reducing motility: larval movement was essentially halted, although, unlike the five samples above, the effect took longer, >3000 min, and/or was not fully sustained throughout the 72 h period (Fig. 5B). The remaining two of the 13 high-interest samples were pure compounds (M1018 – emethacin B, M1025 – epicoccin E). Sample M1018 showed almost complete inhibition of motility from the very start of the run (Fig. 5A). Sample M1025 reduced motility to nearly 0% within ∼3700 min, although larval motility rebounded and was recorded in the 50–75% range before the end of the run (Fig. 5B). This study validates the wMicroTracker system as an economical and high-throughput option for testing large suites of natural products against A.cantonensis, adds to the short list of diverse parasites for which it has been validated and highlights the value of A.cantonensis and Hawaiian fungi for discovery of new anthelmintics.

Parasitology. 2022 Mar 3;1-28. doi: 10.1017/S0031182022000191.
Randi L. Rollins, Mallique Qader, William L. Gosnell, Cong Wang, Shugeng Cao and Robert H. Cowie.
