
There are about 13 parasitic infections that are responsible for significant morbidity and mortality but have not received the attention they deserve; thus, they are now known as “neglected tropical diseases” (NTDs). Most of the helminth parasites are showing resistance to available synthetic drugs. Furthermore, these drugs have side effects and are not easily available to the local population. In Cameroon, the use of herbal medicines for the treatment of several ailments including helminthiases is very common. It is therefore of paramount importance to rapidly screen medicinal plants used by the local population to treat helminthiases in order to justify their usage by the local population. This study was aimed at evaluating the anthelminthic activities of Lophira lanceolate, a medicinal plant used in traditional medicine to treat many diseases, using an automated high-throughput method. Most conventional in vitro screening methods for parasitic worms have had limitations in that most of them are time-consuming to perform and/or lack repeatability/ reproducibility. The WMicroTracker (Phylumtech, Argentina), is rapid, automated high-throughput equipment used for the evaluation of the antihelminthic activity of drugs by sending infrared beams to worms and recording the movement of worms. Heligmosomoides polygyrus is a parasitic nematode used as a model for the evaluation of the antihelminthic activity of drugs.
Methods
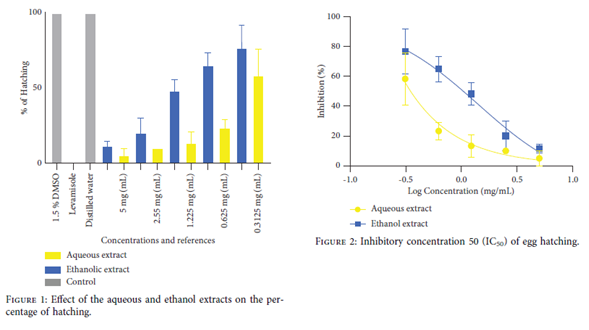
Egg-Hatching Test. 100 µL of the embryonated egg suspension containing 60 eggs was incubated with 100 µL of various concentrations of extracts, levamisole, and 3% DMSO in 96-well cell culture round bottom microtitre plates. The 96-well microplates were incubated for 24 h at 25°C at a relative humidity (RH) of 78% in WMicroTracker, where the worm movements were measured by using WMicroTracker every 30 minutes. Three independent experiments for the ethanol and aqueous extracts were carried out, and the average results were recorded. The inhibition percentages of hatching of each sample were determined at the end of incubation, using the average movement over 24 h in the presence of test samples with the extract, compared with the controls, DMSO 1.5% and levamisole
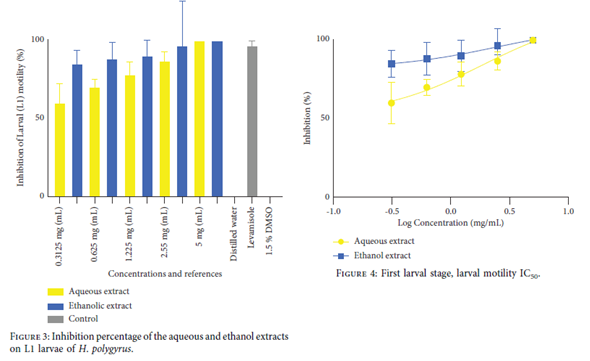
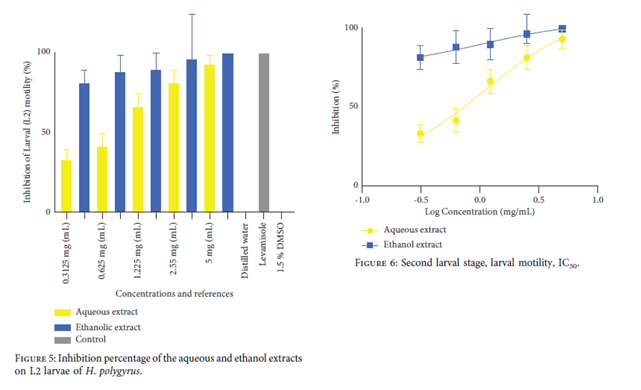
Larval Motility Assays. For the efects of the extracts on L1 and L2 larvae, the same procedure described above was followed. Briefly, 100 µL of the L1 larval stage suspension containing 50 L1 larvae was incubated with 100 µL of various concentrations of extracts, levamisole, and 1.5% DMSO in 96-well cell culture round-bottom microtitre plates. The 96-well microtitre plates were incubated in WMicroTracker at 25°C for 24 h; the worm movements were recorded every 30 min. The larval motility activity was determined by calculating the inhibition percentages. The same method was used to test the activity of active compounds on the L2 larvae.
Results
The highest percentage of hatching for the extracts was observed with the ethanol extract at concentrations of 0.3125 mg/mL and 0.625 mg/mL with hatching percentages of 65±8.6% and 76.66±15.2%, respectively (Figure 1). The ethanol extract exhibited a stronger inhibitory power on the hatching rate of parasites (Figure 2). The aqueous and ethanol extracts of L. lanceolata showed larvicidal activity on the L1 larvae (Figure 3 and 4) with an IC50 of 1.85 mg/mL and 2.4 mg/mL, respectively, as well as on the L2 larvae (Figure 5 and 6) with IC50 values of 1.08 mg/mL and 1.02 mg/mL, respectively. The extracts of L. lanceolata possessed larvicidal properties, with the ethanol extract being the most effective one. The results obtained in our study scientifically validate the use of L. lanceolata in fighting against helminthes in Cameroonian folk medicine. However, in vivo and toxicity tests are necessary to assess its activity and safety.
Journal of Tropical Medicine. Volume 2023, Article ID 9504296, doi: 0.1155/2023/9504296.
Yamssi Cédric, Noumedem A C Nadia, Simeni N S Raoul, Samuel Berinyuy, Mounvera A Azizi, Tientcheu N J Sandra, Ngouyamsa N A Sidiki, and Vincent K Payne.
