
Parkinson’s disease (PD) is a progressive neurodegenerative disorder for which there is no successful prevention or intervention. The pathological hallmark for PD involves the self-assembly of functional Alpha-Synuclein (αS) into non-functional amyloid structures. One of the potential therapeutic interventions against PD is the effective inhibition of αS aggregation. Using an array of biophysical, cellular, in vivo assays and mutation studies, we have identified SK-129, a potent antagonist of αS aggregation in both in vitro and in vivo PD models.
Methods
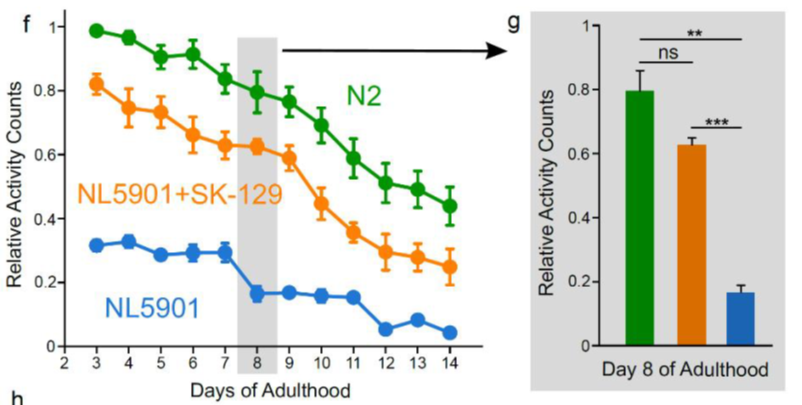
Effect of SK-129 on αS aggregation mediated PD phenotypes in an in vivo model.The antagonist activity of SK-129 against αS aggregation was tested in vivo using a C elegans-based PD model (NL5901). The NL5901 strain expresses WT αS-YFP in the body wall muscle cells and PD phenotypic readouts include a decline in motility during aging. The NL5901 strain was treated with 15 μM SK-129 at the larval stage and incubated with and without SK-129 for nine days.
Results
The motility rate of the NL5901 strain decreases during the aging process as a consequence of αS inclusions. We utilized a newly developed WMicroTracker ARENA plate reader to measure the locomotion (overall activity counts) of NL5901 in the absence and presence of SK-129. The overall activity of NL5901 displayed a gradual decline in the activity in comparison to the WT model of C elegans (N2) (Fig. 5f); however, NL5901 treated with 15 μM SK-129 at the larval stage resulted in a significant improvement in the overall activity (Fig. 5f). The overall activity of NL590 treated with SK-129 was closer to N2 (Fig. 5f).
Nat Commun. 2022 Apr 27;13(1):2273. doi: 10.1038/s41467-022-29724-4.
Jemil Ahmed, Tessa C Fitch, Courtney M Donnelly, Johnson A Joseph, Tyler D Ball, Mikaela M Bassil, Ahyun Son, Chen Zhang, Aurélie Ledreux, Scott Horowitz, Yan Qin, Daniel Paredes, Sunil Kumar.
