
Chagas’ disease or American trypanosomiasis is a neglected tropical disease (NTDWHO), which is endemic in Latin America but has spread in recent decades to nonendemic regions owing to the migration of unaware infected people. Its etiological agent is the homoflagellated protozoan parasite Trypanosoma cruzi (T. cruzi), whose metacyclic trypomastigote form is transmitted to mammalian host by blood-sucking triatomine bugs. Available chemotherapy is based on Benznidazole and Nifurtimox, two drugs developed more than 50 years ago, which proved to be toxic, require a long treatment as compared to the rate of disease progression, and often develop drug resistance. Although many natural and synthetic compounds have been assayed for activity against T. cruzi, very few have successfully progressed through clinical trials. In the current work, a new series of five [VVO(IN-2H)(L-H)] compounds, where L are 8-hydroxyquinoline derivatives L0-L4 and IN is the new tridentate ligand, were synthesized and fully characterized. The biological activity of the whole series of oxidovanadium compounds and the ligands was evaluated against epimastigote and trypomastigote forms of T. cruzi. Finally, the toxicity of the hit compound was evaluated using Caenorhabditis elegans (C. elegans), a nematode model organism that is used for drugs screening as a simplified proxy of animal toxicity.
Methods
The wild-type C. elegans Bristol strain N2 and Escherichia coli (E. coli) OP50 strain were obtained from the Caenorhabditis Genomics Center (Minneapolis, MN, USA). Worms were maintained under standard conditions at 20 ºC on Nematode Growth Media (NGM) agar plates seeded with E. coli OP50 as a source of food. The method used to assess toxicity is based on worm motility. Briefly, the locomotor activity recording system, WMicrotrackerTM ONE (PhylumTech, Santa Fe, Argentina) detects infrared microbeam interruptions. Synchronized L4 C. elegans worms were removed from culture plates and washed three times with M9 buffer (3 g KH2PO4; 6 g Na2HPO4; 5 g NaCl; 1 mL 1MMgSO4 per liter) by centrifugation at 1000 g. Worms in M9 1% DMSO were plated in 96-well flat microtiter plates (Deltalab, Barcelona, Spain). Approximately 70 worms per well were seeded in 60 µL M9 buffer containing 1% DMSO and their basal movement was measured for 30 min to normalize the movement activity for each well at the beginning of the assay. Then, compound [VVO(IN-2H)(L2-H)] dissolved in M9 buffer 1% DMSO was added to a final concentration range of 6.5–100 µM in a final volume of 100 µL per well. Vehicle alone (1% DMSO) and the anthelmintic drug ivermectin at 2 µM were used as used controls. The motility using WMicrotrackerTM ONE was measured for 18 h. Motility of worms with vehicle only after 18 h was considered for reference as 100% motility. Four replicas were performed for each concentration in 96-well plates. Three biological replicas were performed.
Results
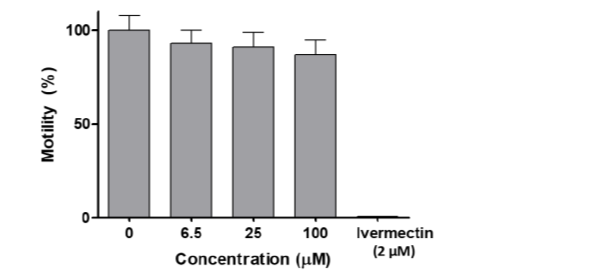
The dose–response study on C. elegans showed that [VVO(IN-2H)(L2-H)] did not affect worm motility (Figure 9). At a concentration as high as 100 µM, which is the maximal solubility of the compound at 1% DMSO, there was a marginal decrease in motility after 18 h, which was not statistically significant (unpaired t test). In contrast, the known anthelmintic drug ivermectin, used as a positive control, reduced motility to zero at 2 µM (Figure 9). The worms treated with compound [VVO(IN-2H)(L2-H)] at 100 µM or with vehicle only were recovered from wells and seeded on NGM plates and food was added to assess viability and development. Worms treated at 100 µM were all alive and showed normal development (L4–adult worm–egg–L1). [VVO(IN-2H)(L2-H)] did not significantly affect motility after 18 h treatment in the concentration range used (6.5–100 µM). In addition, neither viability nor development were affected after treatment. These results indicate that this compound is innocuous for this animal invertebrate model that allows acute toxicity and developmental and reproductive toxicity (DART) to be examined.
Heteroleptic Oxidovanadium(V) Complexes with Activity against Infective and Non-Infective Stages of Trypanosoma cruzi. Molecules 2021, 26, 5375.
Scalese, G.; Machado, I.; Salinas, G.; Pérez-Díaz, L.; Gambino, D.
