
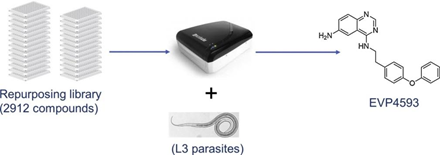
Parasitic nematodes continue to cause significant economic losses in livestock globally. Given the limited number of anthelmintic drugs on the market and the currently increasing drug resistance, there is an urgent need for novel anthelmintics. The assessment of motility is considered to be the current gold standard for measuring drug effectiveness for helminth parasites in vitro. Most motility assays of anthelmintic activity for parasitic nematodes are laborious and low throughput, and therefore not suitable for screening large compound libraries. Therefore, using a WMicrotracker One instrument, we established a practical, automated and low-cost whole-organism motility assay against ex sheathed L3 stages (xL3s) of the ruminant parasite Cooperia oncophora, and screened a repurposing library comprising 2745 molecules. Fourteen known anthelmintics contained in this library were picked up in this blind screen, as well as four novel hits: thonzonium bromide, NH125, physostigmine sulfate, and EVP4593. The four hits were also active against xL3s of Ostertagia ostertagi, Haemonchus contortus and Teladorsagia circumcincta using the same assay.
Material and Methods
The anthelmintic assays were performed in sterile 96-well flat bottom microplates. The repurposing library was screened using a blinded screening approach. One μL of individual compound stock solution (10mM in DMSO) from the library was added into 99 μL of RPMI-1640 in the 96-well plate and arrayed in duplicate. The collectedxL3s of C.oncophora in RPMI-1640 were adjusted approximately to 800larvae/mL. Then 80 xL3s in 100 μL of RPMI-1640 were transferred to each well. The final compound concentration was 50 μM containing 0.5% DMSO (v/v). Thus, 0.5% DMSO (four replicates) was used as a solvent control, while 50 μM levamisole (four replicates) was used a positive control. The plate was agitated (300 rpm/min) for 10 min and incubated at 37 °C in a humidified incubator with a 5% CO2 atmosphere for 8 h. Then the media in the wells were gently pipetted up and down 5 times using a multichannel pipette to stimulate the worms. Subsequently, the plate was placed into a WMicrotracker ONE device. Then the worms were allowed to habituate for 5 min in the (dark) chamber of the WMicrotracker, followed by incubation and motility monitoring for 3 h at 20 °C. The motility of worms in each well was measured every 30 min. A darkness/light (1 h/1 h) cycle in the WMicrotracker was used as a stimulus during tracking. The percentage of the average movement over 3 h of exposure to test compounds, compared with the DMSO control, was used to estimate the relative anthelmintic activity. The same protocol was used to test the activity of C.oncophora hits on xL3s of O.ostertagi, H.contortus and T.circumcincta.
Results
Of all 2735 compounds in the repurposing library, 18 reduced worms motility by ≥ 70% in both the primary and secondary screen, and were considered as hits. After their structures were revealed (up to that point the screening was blind), 14 turned out to be known anthelmintic, and were not considered for further studies. The other four hits are not known as anthelmintic agents; thonzonium bromide, NH125, physostigmine sulfate and EVP4593. Their EC50 values were determined, and EVP4593 showed the most potent activity. The four hits were also active against xL3s of three other ruminant parasites: Ostertagia ostertagi, Haemonchus contortus and Teladorsagia circumcincta (Table 1). Screening a repurposing compound library led to four new hits. One of these (EVP4593) demonstrated promising properties as an anthelmintic.
Vet Parasitol. 2019 Jan;265:15-18. doi: 10.1016/j.vetpar.2018.11.014. Epub 2018 Dec 7.
Maoxuan Liu, Bart Landuyt, Hugo Klaassen, Peter Geldhof, Walter Luyten
